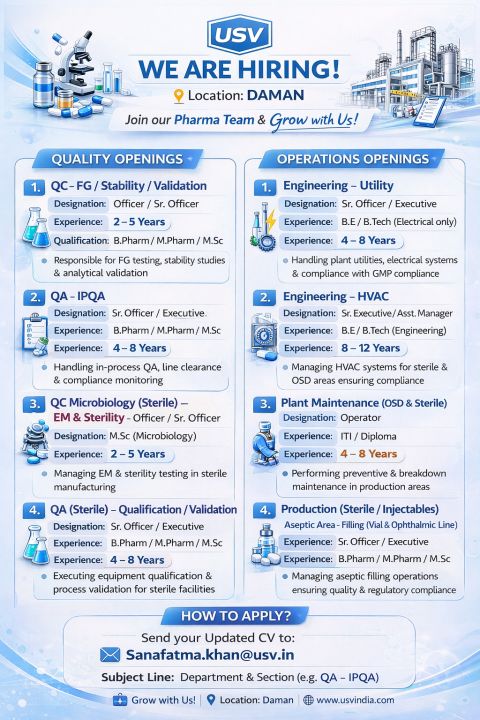
Quality Assurance Executive – Quality Assurance Sterile Qualification and Validation | USV Private Limited | Daman, Gujarat, India
USV Private Limited is seeking a Quality Assurance Executive for its sterile pharmaceutical operations in Daman, Gujarat, India. This full-time role is suitable for professionals with 4 to 8 experience in quality assurance, validation, and qualification activities within the Pharmaceutical industry in India. The position ensures compliance with GMP guidelines, validation protocols, and regulatory documentation standards.
Location
Daman, Gujarat, India
Employment Type
Full-time
Experience Required
4 to 8 experience in pharmaceutical quality assurance
Compensation & Benefits
Competitive pharmaceutical industry compensation with structured career growth.
About USV Private Limited
USV Private Limited is a reputed pharmaceutical organization committed to delivering high-quality medicines through strong quality systems and validated manufacturing processes.
Position Overview
The Quality Assurance Executive ensures product quality through validation, qualification, and compliance monitoring activities.
Key Responsibilities
- Review validation and qualification documents.
- Ensure GMP compliance across departments.
- Support equipment and process validation.
- Maintain QA documentation systems.
- Participate in internal audits.
- Monitor deviations and CAPA activities.
- Review batch manufacturing records.
- Ensure regulatory compliance.
- Coordinate cross-functional QA activities.
- Support inspection readiness.
Required Skills & Competencies
- Pharmaceutical QA expertise.
- Validation and qualification knowledge.
- GMP compliance understanding.
- Documentation review skills.
- Analytical thinking ability.
- Regulatory awareness.
- Attention to detail.
- Audit preparation capability.
- Strong coordination skills.
Educational Qualification
BPharm MPharm MSc
About the Project & Industry
This role supports sterile pharmaceutical manufacturing aligned with global regulatory and compliance frameworks.
Why Join USV Private Limited?
Employees gain exposure to advanced pharmaceutical quality systems and validation practices.
How to Apply
Sanatamfa.khan@usv.in