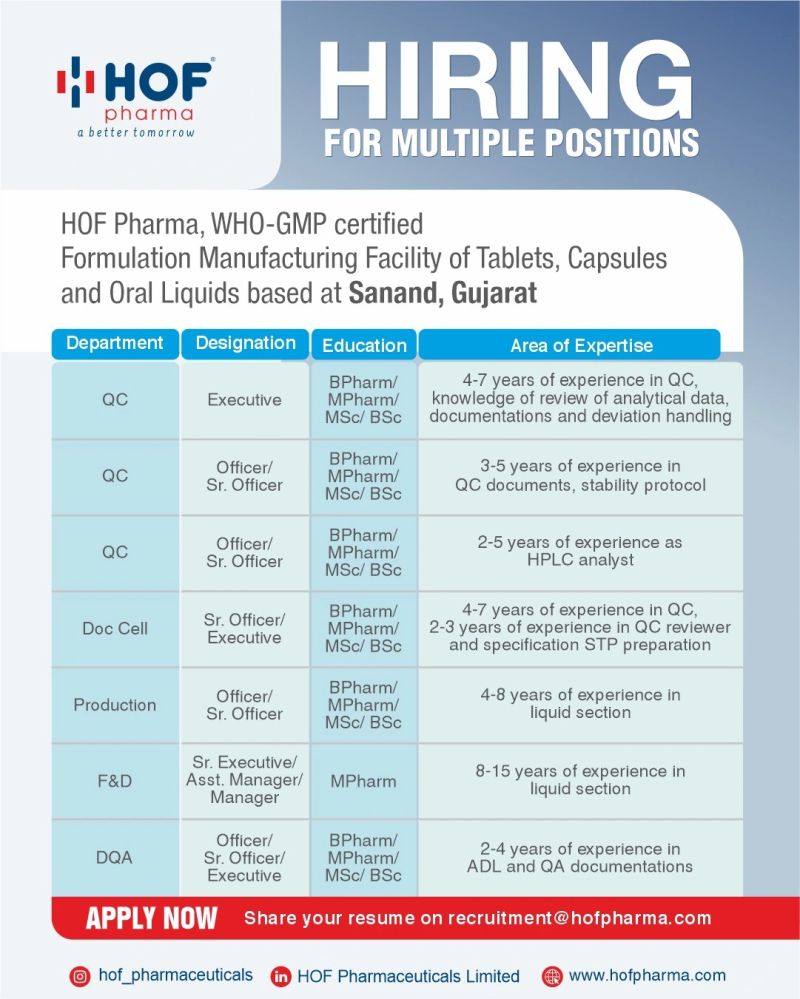
Company Introduction
HOF Pharma maintains regulated quality systems supporting pharmaceutical manufacturing and product release.
Core Responsibilities
- Review analytical data packages against approved specifications.
- Prepare audit response files using controlled QA formats.
- Maintain deviation registers and investigation documentation.
- Check batch records for completeness prior to disposition.
- Update quality logs and compliance trackers.
Skills & Technical Exposure
- Deviation handling documentation.
- Quality management system records.
- Batch record review checklists.
- Audit response documentation.
- Controlled QA registers.
Experience Context
- Work is performed within QA offices connected to production and QC.
- Documentation volume spans multiple commercial batches monthly.
- Without this role, product disposition remains unsupported.
How to Apply
recruitment@hofpharma.com