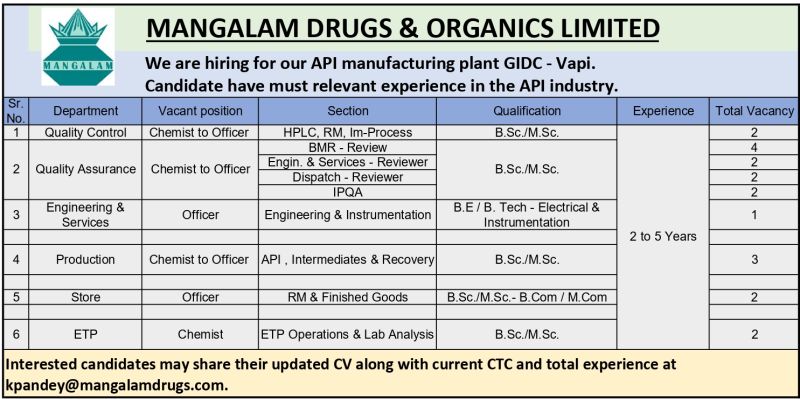
Company Introduction
Mangalam Drugs & Organics Limited operates regulated pharmaceutical facilities supplying APIs to domestic and international markets.
Core Responsibilities
- Review batch manufacturing records, deviation notes, and investigation reports using approved QA formats.
- Inspect IPQA documents, line clearance records, and area checklists on production floors.
- Verify equipment cleaning records through logbooks, cleaning validation files, and checklists.
- Check compliance documents against regulatory guidelines using SOP binders and audit tools.
- Maintain controlled document registers and archival files within QA storage systems.
Skills & Technical Exposure
- Batch manufacturing records, deviation forms, and investigation templates.
- IPQA checklists, line clearance formats, and area inspection logs.
- SOP manuals, quality manuals, and change control documents.
- Audit checklists, compliance trackers, and CAPA records.
- Document control registers and archival systems.
Experience Context
- Work setting spans QA offices and API manufacturing production blocks.
- Document volume covers multiple API batches and validation cycles.
- Without this role, regulatory readiness weakens and compliance gaps remain unresolved.
How to Apply
Interested candidates may send their CV to kpandey@mangalamdrugs.com