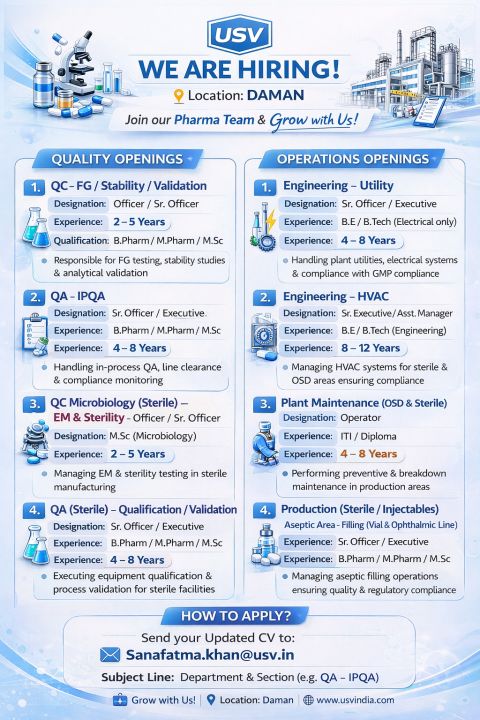
QC Officer – Quality FG Stability and Validation | USV Private Limited | Daman, Gujarat, India
USV Private Limited is hiring a QC Officer for Quality FG Stability and Validation at its Daman, Gujarat, India pharmaceutical facility. This full-time role is ideal for professionals with 2 to 5 years experience in finished goods testing, stability studies, and validation activities within the Pharmaceutical industry in India. The position focuses on maintaining product quality through analytical testing, ensuring compliance with GMP regulations, supporting validation protocols, and contributing to regulatory-ready laboratory operations. Candidates will collaborate closely with quality assurance and production teams to ensure consistent pharmaceutical quality standards.
Location
Daman, Gujarat, India
Employment Type
Full-time
Experience Required
2 to 5 years experience in pharmaceutical quality control and stability testing
Compensation & Benefits
Compensation aligned with Pharmaceutical industry in India standards along with professional laboratory exposure and career growth opportunities.
About USV Private Limited
USV Private Limited is a reputed organization in the Pharmaceutical industry in India known for quality-driven manufacturing and regulatory-compliant healthcare product development.
Position Overview
The QC Officer will manage finished goods testing, stability programs, and validation activities to ensure product compliance with regulatory requirements.
Key Responsibilities
- Perform finished goods analytical testing.
- Conduct stability study evaluations.
- Support validation and qualification activities.
- Maintain laboratory documentation.
- Ensure GMP compliance in QC operations.
- Review analytical data and reports.
- Coordinate with QA for investigations.
- Maintain instrument calibration records.
- Assist audit and inspection readiness.
- Ensure adherence to SOP requirements.
Required Skills & Competencies
- Quality control laboratory expertise.
- Stability testing knowledge.
- Validation documentation skills.
- GMP compliance understanding.
- Analytical instrumentation knowledge.
- Data analysis capability.
- Attention to detail.
- Regulatory compliance awareness.
- Team coordination skills.
Educational Qualification
BPharm MPharm MSc
About the Project & Industry
This role supports pharmaceutical quality systems ensuring regulatory compliance and product stability within the Pharmaceutical industry in India.
Why Join USV Private Limited?
Opportunity to work in a compliance-driven pharmaceutical environment with advanced laboratory exposure.
How to Apply
Sanatamfa.khan@usv.in