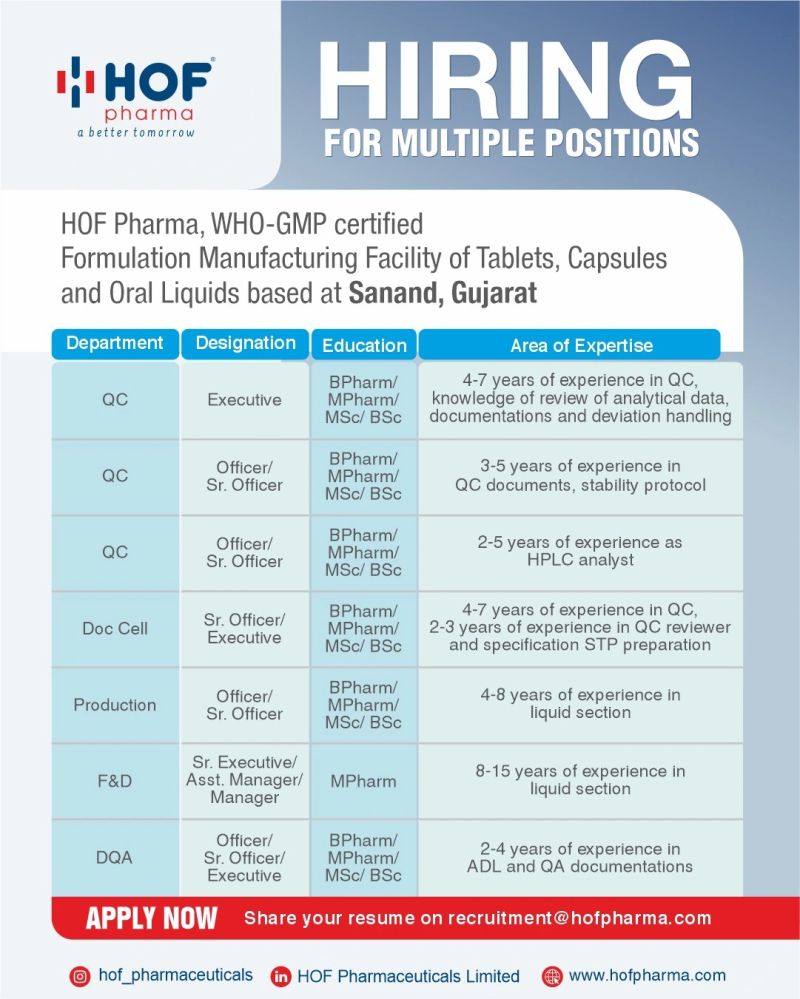
Company Introduction
HOF Pharma develops and manufactures oral solid and liquid dosage forms under regulated quality systems.
Core Responsibilities
- Prepare controlled SOP drafts, specifications, and master documents.
- Compile analytical review files using test reports and approved formats.
- Update document indices, version trackers, and archival registers.
- Check completed records for completeness against approved templates.
- Assemble regulatory-ready documentation sets for internal review.
Skills & Technical Exposure
- Standard operating procedures and specification formats.
- Document control logs and issuance registers.
- Change history sheets and archival systems.
- Electronic document management systems.
- Controlled master list documentation.
Experience Context
- Work is performed in documentation cells supporting QC and QA teams.
- Document sets may span hundreds of controlled pages per product.
- Absence of this role results in audit gaps and untraceable records.
How to Apply
recruitment@hofpharma.com